 
This information can be crucial for understanding the properties and behavior of the compound, as well as for identifying it. Percent composition tells us the relative amounts of each element in a chemical compound. This is because naturally occurring carbon is a mixture of the isotopes 12C, 13C and 14C which have masses of 12 Da, 13.003355 Da, and 14.003242 Da respectively. For example, the standard atomic weight of carbon is 12.011 g/mol, not 12.00 g/mol. When calculating the molar mass in our Percent Composition Calculator we use the chemical elements’ atomic weights based on the isotopic composition of the elements. So, in case of carbon-12 we can say that its molecular (actually atomic) weight is 12 g/mol. Since, by definition, one mole is the amount of substance in 12 grams of carbon-12, the molecular weight can be expressed by the same number but in units g/mol instead of daltons. It is the mass of 1 mole of the substance or 6.022×10 23 particles, expressed in grams (g/mol). The molar mass of a chemical compound is defined as the mass of a sample of that compound divided by the amount of substance in that sample, measured in moles. 
Parentheses ( ), square brackets and braces (curly brackets) \times 100\% $$ Indices should be entered as normal numbers after the appropriate elements or groups, e.g. The chemical formula should be entered using the upper case for the first character in the element’s name and the lower case for the second character (compare: Co – cobalt and CO – carbon monoxide). PE = (|theoretical value – experimental value| \ theoretical value) x 100%.Īll three versions of the formula mean the exact same thing - it’s just different wording.This online Percent Composition Calculator finds the elemental composition as well as the percentage distribution of the number of atoms and their masses for a molecule given its chemical formula. 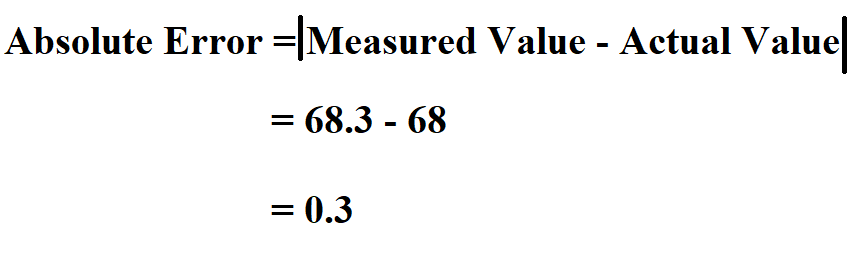
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
